.avif)
.png)
Health science sales outsourcing allows life sciences companies to accelerate market entry, reduce fixed costs, and access specialized expertise by partnering with third-party sales teams. This guide covers the strategic benefits, service models (from full-service to clinical support), critical selection criteria for outsourcing partners, and compliance considerations specific to FDA-regulated medical devices and pharmaceuticals.
Health science companies face unprecedented pressure. Shrinking patent lives, stringent regulatory requirements, and fierce market competition demand faster product launches and leaner operations. Outsourcing sales functions has shifted from a cost-cutting tactic to a strategic accelerator that unlocks specialized expertise and market access.
The medical device outsourcing market was valued at approximately USD 128.8 billion in 2023 and is projected to reach USD 262.3 billion by 2032, growing at a CAGR of 8.2%. According to industry research, 90% of health executives are exploring cost savings by teaming with third-party vendors to achieve operational efficiency.
But here's the thing—outsourcing health science sales isn't just about handing off tasks. It's about selecting the right partner, understanding compliance requirements, and aligning service models with business goals.
What Is Health Science Sales Outsourcing?
Health science sales outsourcing involves partnering with specialized third-party organizations to handle sales activities for medical devices, pharmaceuticals, diagnostics, or biotech products. These partners—often called Contract Sales Organizations (CSOs)—bring trained sales representatives, established healthcare networks, and proven processes.
Unlike traditional hiring, outsourced sales converts fixed overhead into variable costs tied to performance. Companies gain immediate market access without the lengthy recruitment and training cycles that drain resources.
Services range from full-service commercial teams handling territory management and customer relationships to targeted support like clinical education, product demonstrations, or regulatory affairs assistance.
Key Service Models
Different business needs call for different outsourcing approaches:
Why Companies Outsource Health Science Sales
The decision to outsource stems from both strategic opportunities and operational realities. Companies aren't just cutting costs—they're fundamentally rethinking how to compete.
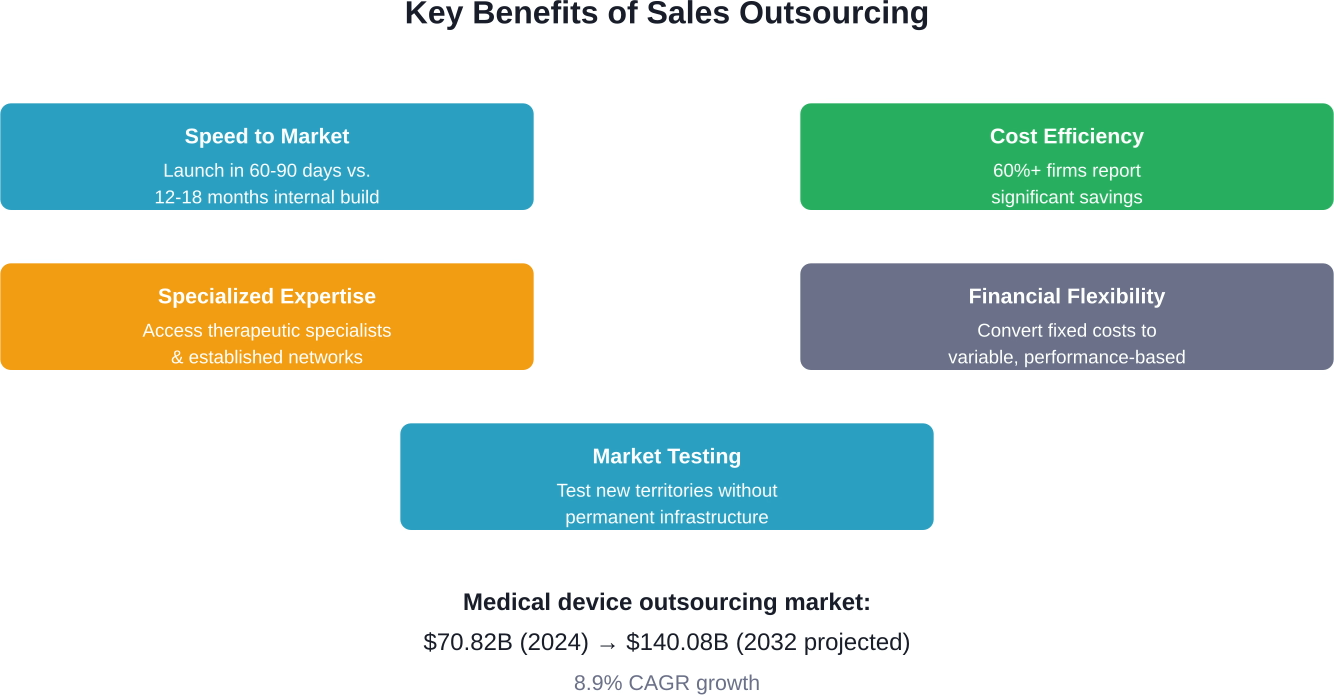
Speed to Market
Product development timelines are ruthless. A delay of even six months can mean millions in lost revenue, especially for medical devices with limited patent protection. Building an internal sales force typically requires 12-18 months. Outsourced teams can launch in 60-90 days.
For companies introducing Class II medical devices—which account for 43% of all medical device types—this speed advantage can determine market leadership versus playing catch-up.
Cost Efficiency and Financial Flexibility
According to competitor content, over 60% of firms report significant savings through sales outsourcing. The economics are straightforward: eliminate recruitment costs, training infrastructure, benefits administration, and sales management overhead.
Variable cost structures matter especially during market uncertainty. If a product underperforms or market conditions shift, companies can adjust outsourced capacity without layoffs or severance packages.
Specialized Expertise
Healthcare sales requires deep therapeutic knowledge, established relationships with key opinion leaders, and credibility within clinical communities. Building this expertise internally takes years.
Outsourcing partners often specialize in specific therapeutic areas—oncology, cardiology, orthopedics—bringing reps who already speak the clinical language and understand prescriber behavior patterns.
Market Testing Without Commitment
Entering a new geographic market or therapeutic segment carries risk. Outsourced sales teams allow companies to test market viability without the fixed costs of establishing permanent operations. If the market responds favorably, companies can scale. If not, they can pivot without painful restructuring.
Build a Stable Health Science Sales Outsourcing Team
Health science sales outsourcing requires product knowledge, structured outreach, and long-term relationship building. NeoWork helps organizations build dedicated remote sales support teams for prospecting, CRM updates, appointment setting, and pipeline coordination. Their differentiators are an industry-leading 91% annualized teammate retention rate and a 3.2% candidate selectivity rate. That stability reduces retraining cycles and supports consistent sales execution in regulated environments.

Ready to Outsource Health Science Sales Support With Long-Term Continuity?
Talk with NeoWork to:
- build a focused remote sales support team
- maintain structured CRM and outreach processes
- scale prospecting without frequent turnover
👉 Connect with NeoWork to structure your health science sales outsourcing model.
Challenges and Considerations
Real talk: outsourcing isn't a magic solution. It introduces complexity that companies must manage deliberately.
Loss of Direct Control
When sales reps don't report directly to your organization, messaging consistency and brand representation become concerns. Outsourced teams serve multiple clients, which can dilute focus during critical launch periods.
Mitigation requires robust service level agreements (SLAs), regular performance reviews, and clear escalation protocols. Companies that treat outsourcing partners as true strategic collaborators—not just vendors—see better alignment.
Compliance and Regulatory Complexity
FDA regulations don't disappear when sales functions are outsourced. Companies remain responsible for ensuring promotional materials comply with regulatory standards, adverse event reporting happens promptly, and sales practices meet industry guidelines.
According to FDA guidance, compliance programs provide instructions to agency staff for obtaining information to fulfill regulatory plans. While these programs don't create legal obligations beyond existing statutes, they establish expectations for oversight.
For outsourcing facilities registered under section 503B of the Federal Food, Drug, and Cosmetic Act, regulatory scrutiny intensifies. FDA warning letters demonstrate the agency's active enforcement against facilities failing to meet compounding and quality standards.
Knowledge Transfer Gaps
Outsourced reps need deep product knowledge to credibly engage healthcare professionals. Inadequate training leads to missed opportunities and damaged relationships. Companies must invest in comprehensive onboarding, even when using external teams.
Cultural and Strategic Misalignment
Some outsourcing partners prioritize volume metrics over relationship quality. Others lack experience in specific therapeutic areas. Mismatched expectations create friction that undermines commercial success.
Choosing the Right Outsourcing Partner
Partner selection determines whether outsourcing accelerates growth or creates headaches. Companies need a systematic evaluation framework.
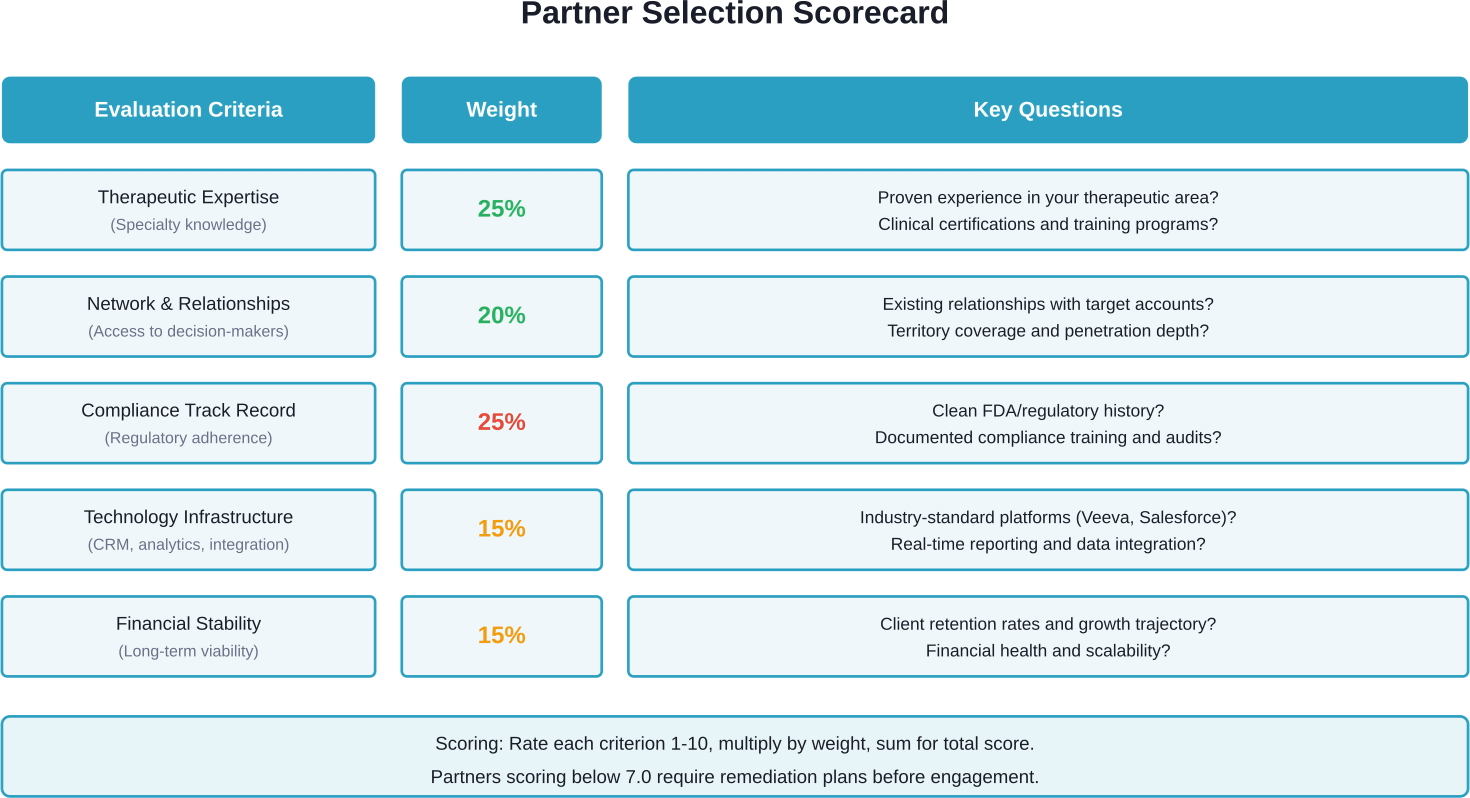
Therapeutic Expertise
Does the partner have demonstrable experience in your therapeutic area? General healthcare sales skills don't transfer seamlessly across specialties. An orthopedic device requires different clinical knowledge than oncology pharmaceuticals.
Request case studies, client references from similar product categories, and evidence of therapeutic training programs. Evaluate whether their reps hold relevant certifications or credentials.
Network and Relationships
Established relationships with key opinion leaders, hospital systems, and group purchasing organizations create immediate credibility. Partners with existing access bypass months of door-opening efforts.
Ask about territory coverage, current client conflicts, and relationship depth with targeted accounts. A partner calling on 200 accounts superficially is less valuable than one with deep relationships across 75 priority targets.
Technology Infrastructure
Modern sales requires sophisticated CRM systems, sample management platforms, and analytics capabilities. According to research on IT technology benefits in clinical trials, outsourcing strategy paired with robust technology improves compliance and quality outcomes.
Evaluate whether partners use industry-standard platforms (Salesforce, Veeva), provide real-time reporting dashboards, and integrate with your existing systems. Data visibility shouldn't decrease just because sales are outsourced.
Compliance Track Record
Request documentation of compliance training programs, audit results, and any regulatory citations. Partners with FDA warning letters or industry violations introduce unacceptable risk.
Ensure the partner maintains current PhRMA Code training, adverse event reporting procedures, and promotional review processes that align with your standards.
Financial Stability and Scale
Can the partner support your growth trajectory? A CSO struggling financially may lack resources for adequate training or technology investments. Conversely, very large organizations may treat smaller clients as low priority.
Review financial statements, client retention rates, and growth projections. Partners experiencing high turnover or client losses signal operational problems.
Critical Compliance Considerations
Regulatory compliance isn't negotiable in health sciences. Outsourcing creates additional complexity that requires proactive management.
FDA Oversight and Responsibility
Companies retain full regulatory responsibility even when sales activities are outsourced. FDA doesn't recognize outsourcing as a defense against promotional violations or adverse event reporting failures.
For facilities registered as outsourcing facilities under section 503B, FDA conducts inspections and issues warning letters when violations occur. Recent enforcement actions demonstrate the agency's commitment to holding both manufacturers and their partners accountable.
Promotional Material Review
All sales materials, presentations, and promotional content require thorough review before use. Establish joint review committees that include regulatory affairs, medical affairs, legal, and partner representatives.
Many companies require partners to submit all materials through formal MLR (Medical Legal Regulatory) review processes, with documented approvals before distribution.
Adverse Event Reporting
Sales reps often receive the first reports of product problems or adverse events. Outsourced teams must understand reporting obligations, timelines, and escalation procedures.
Build these requirements into training programs and audit them regularly. A single missed adverse event report can trigger FDA enforcement actions and legal liability.
Off-Label Promotion Risks
Healthcare professionals frequently ask about off-label uses. Reps must navigate these conversations carefully, providing only FDA-approved information while directing clinical questions to medical affairs.
Partners need clear protocols for handling off-label inquiries, documented training on permissible responses, and regular compliance monitoring.
Emerging Trends Reshaping Health Science Outsourcing
The outsourcing landscape continues evolving rapidly. Several trends are reshaping how companies approach these partnerships.
Chronic Disease Burden Driving Demand
According to WHO and UN data, chronic diseases (non-communicable diseases) are projected to account for 70% of all deaths globally by 2030. This creates sustained demand for new medical products with accelerated launch timelines—precisely the scenario where outsourced sales provides maximum advantage.
Digital and Hybrid Engagement Models
Telehealth adoption fundamentally changed healthcare sales. According to AMA data, while nearly 65-90% of physicians had the capability to provide telehealth in 2021, the actual consistent usage by physicians for consultations stabilized at around 25% of all patient visits by 2023-2024, though over 80% maintain the offering. Outsourcing partners increasingly blend traditional field sales with digital engagement capabilities.
Some 40% of patients indicate they'll continue using virtual care alternatives. This shift requires sales teams comfortable with virtual demonstrations, remote training, and digital relationship-building.
Value-Based Contracting Expertise
Healthcare systems increasingly demand outcomes-based pricing and risk-sharing arrangements. Sales teams need sophisticated understanding of health economics, real-world evidence, and value proposition development.
Outsourcing partners with dedicated value and access specialists help companies navigate these complex negotiations without building internal health economics teams.
Specialized Therapeutic Niches
As therapeutic areas become more specialized—gene therapies, precision oncology, rare diseases—sales expertise becomes harder to build internally. Outsourcing partners who invest in ultra-specialized training and certification provide access to capabilities that would be prohibitively expensive to develop in-house.
Implementation Best Practices
Successful outsourcing requires more than selecting the right partner. Implementation discipline determines whether the relationship delivers value.
Start with Pilot Programs
Rather than committing to large-scale outsourcing immediately, test the relationship through limited geography or specific product pilots. This allows both parties to refine processes, identify gaps, and build trust before scaling.
Define clear success metrics for the pilot—sales targets, relationship quality indicators, compliance audit results—and use these to guide expansion decisions.
Invest in Comprehensive Onboarding
Treat outsourced reps like internal employees during onboarding. Provide the same clinical training, product knowledge development, and company culture immersion.
Companies that skimp on training see it reflected in rep credibility and sales results. The investment pays dividends through better customer interactions and faster ramp times.
Establish Clear Communication Cadence
Weekly or biweekly sync meetings between internal teams and outsourced partners prevent misalignment. These should cover pipeline development, competitive intelligence, customer feedback, and tactical adjustments.
Don't rely solely on formal reporting. Regular informal communication builds relationships and surfaces issues before they become problems.
Create Joint Success Metrics
Align incentives by measuring what matters to both parties. Beyond sales volume, consider metrics like customer satisfaction scores, compliance audit results, relationship depth indicators, and strategic account penetration.
When partners share accountability for outcomes beyond just revenue, collaboration improves and short-term thinking decreases.
Conduct Regular Performance Reviews
Quarterly business reviews should assess performance against SLAs, identify improvement opportunities, and recalibrate priorities. These aren't punitive exercises—they're collaborative sessions to optimize the partnership.
Include recognition of successes alongside constructive feedback. Partners who feel valued as strategic collaborators deliver better results than those treated as interchangeable vendors.
Conclusion: Strategic Outsourcing Requires Strategic Planning
Health science sales outsourcing has matured from a tactical cost-reduction tool into a strategic capability that accelerates market entry, provides specialized expertise, and creates financial flexibility. The medical device outsourcing market's projected growth to USD 140.08 billion by 2032 reflects this fundamental shift.
But success isn't automatic. Companies that approach outsourcing strategically—selecting partners based on therapeutic expertise and compliance track records, investing in comprehensive training, establishing clear metrics, and treating partners as true collaborators—achieve significantly better results than those simply seeking the lowest cost provider.
The regulatory environment adds complexity that can't be delegated. FDA oversight, promotional compliance, and adverse event reporting remain the manufacturer's responsibility regardless of outsourcing arrangements. Partners must demonstrate not just sales capability, but regulatory sophistication and commitment to compliance.
For companies facing accelerated launch timelines, resource constraints, or expansion into new therapeutic areas, outsourcing provides a proven path forward. The key is approaching these partnerships with the same strategic rigor applied to internal capability development.
Ready to explore health science sales outsourcing for your organization? Start by conducting a thorough needs assessment identifying which sales functions deliver maximum value when outsourced, developing detailed partner selection criteria weighted toward therapeutic expertise and compliance, and designing pilot programs that test capabilities before full-scale commitments. The right partner can become a competitive advantage—choose deliberately.
Frequently Asked Questions
Topics
Health Science Sales Outsourcing Guide 2026
Health science sales outsourcing allows life sciences companies to accelerate market entry, reduce fixed costs, and access specialized expertise by partnering with third-party sales teams. This guide covers the strategic benefits, service models (from full-service to clinical support), critical selection criteria for outsourcing partners, and compliance considerations specific to FDA-regulated medical devices and pharmaceuticals.
Health science companies face unprecedented pressure. Shrinking patent lives, stringent regulatory requirements, and fierce market competition demand faster product launches and leaner operations. Outsourcing sales functions has shifted from a cost-cutting tactic to a strategic accelerator that unlocks specialized expertise and market access.
The medical device outsourcing market was valued at approximately USD 128.8 billion in 2023 and is projected to reach USD 262.3 billion by 2032, growing at a CAGR of 8.2%. According to industry research, 90% of health executives are exploring cost savings by teaming with third-party vendors to achieve operational efficiency.
But here's the thing—outsourcing health science sales isn't just about handing off tasks. It's about selecting the right partner, understanding compliance requirements, and aligning service models with business goals.
What Is Health Science Sales Outsourcing?
Health science sales outsourcing involves partnering with specialized third-party organizations to handle sales activities for medical devices, pharmaceuticals, diagnostics, or biotech products. These partners—often called Contract Sales Organizations (CSOs)—bring trained sales representatives, established healthcare networks, and proven processes.
Unlike traditional hiring, outsourced sales converts fixed overhead into variable costs tied to performance. Companies gain immediate market access without the lengthy recruitment and training cycles that drain resources.
Services range from full-service commercial teams handling territory management and customer relationships to targeted support like clinical education, product demonstrations, or regulatory affairs assistance.
Key Service Models
Different business needs call for different outsourcing approaches:
Why Companies Outsource Health Science Sales
The decision to outsource stems from both strategic opportunities and operational realities. Companies aren't just cutting costs—they're fundamentally rethinking how to compete.
Speed to Market
Product development timelines are ruthless. A delay of even six months can mean millions in lost revenue, especially for medical devices with limited patent protection. Building an internal sales force typically requires 12-18 months. Outsourced teams can launch in 60-90 days.
For companies introducing Class II medical devices—which account for 43% of all medical device types—this speed advantage can determine market leadership versus playing catch-up.
Cost Efficiency and Financial Flexibility
According to competitor content, over 60% of firms report significant savings through sales outsourcing. The economics are straightforward: eliminate recruitment costs, training infrastructure, benefits administration, and sales management overhead.
Variable cost structures matter especially during market uncertainty. If a product underperforms or market conditions shift, companies can adjust outsourced capacity without layoffs or severance packages.
Specialized Expertise
Healthcare sales requires deep therapeutic knowledge, established relationships with key opinion leaders, and credibility within clinical communities. Building this expertise internally takes years.
Outsourcing partners often specialize in specific therapeutic areas—oncology, cardiology, orthopedics—bringing reps who already speak the clinical language and understand prescriber behavior patterns.
Market Testing Without Commitment
Entering a new geographic market or therapeutic segment carries risk. Outsourced sales teams allow companies to test market viability without the fixed costs of establishing permanent operations. If the market responds favorably, companies can scale. If not, they can pivot without painful restructuring.

Build a Stable Health Science Sales Outsourcing Team
Health science sales outsourcing requires product knowledge, structured outreach, and long-term relationship building. NeoWork helps organizations build dedicated remote sales support teams for prospecting, CRM updates, appointment setting, and pipeline coordination. Their differentiators are an industry-leading 91% annualized teammate retention rate and a 3.2% candidate selectivity rate. That stability reduces retraining cycles and supports consistent sales execution in regulated environments.

Ready to Outsource Health Science Sales Support With Long-Term Continuity?
Talk with NeoWork to:
- build a focused remote sales support team
- maintain structured CRM and outreach processes
- scale prospecting without frequent turnover
👉 Connect with NeoWork to structure your health science sales outsourcing model.
Challenges and Considerations
Real talk: outsourcing isn't a magic solution. It introduces complexity that companies must manage deliberately.
Loss of Direct Control
When sales reps don't report directly to your organization, messaging consistency and brand representation become concerns. Outsourced teams serve multiple clients, which can dilute focus during critical launch periods.
Mitigation requires robust service level agreements (SLAs), regular performance reviews, and clear escalation protocols. Companies that treat outsourcing partners as true strategic collaborators—not just vendors—see better alignment.
Compliance and Regulatory Complexity
FDA regulations don't disappear when sales functions are outsourced. Companies remain responsible for ensuring promotional materials comply with regulatory standards, adverse event reporting happens promptly, and sales practices meet industry guidelines.
According to FDA guidance, compliance programs provide instructions to agency staff for obtaining information to fulfill regulatory plans. While these programs don't create legal obligations beyond existing statutes, they establish expectations for oversight.
For outsourcing facilities registered under section 503B of the Federal Food, Drug, and Cosmetic Act, regulatory scrutiny intensifies. FDA warning letters demonstrate the agency's active enforcement against facilities failing to meet compounding and quality standards.
Knowledge Transfer Gaps
Outsourced reps need deep product knowledge to credibly engage healthcare professionals. Inadequate training leads to missed opportunities and damaged relationships. Companies must invest in comprehensive onboarding, even when using external teams.
Cultural and Strategic Misalignment
Some outsourcing partners prioritize volume metrics over relationship quality. Others lack experience in specific therapeutic areas. Mismatched expectations create friction that undermines commercial success.
Choosing the Right Outsourcing Partner
Partner selection determines whether outsourcing accelerates growth or creates headaches. Companies need a systematic evaluation framework.
Therapeutic Expertise
Does the partner have demonstrable experience in your therapeutic area? General healthcare sales skills don't transfer seamlessly across specialties. An orthopedic device requires different clinical knowledge than oncology pharmaceuticals.
Request case studies, client references from similar product categories, and evidence of therapeutic training programs. Evaluate whether their reps hold relevant certifications or credentials.
Network and Relationships
Established relationships with key opinion leaders, hospital systems, and group purchasing organizations create immediate credibility. Partners with existing access bypass months of door-opening efforts.
Ask about territory coverage, current client conflicts, and relationship depth with targeted accounts. A partner calling on 200 accounts superficially is less valuable than one with deep relationships across 75 priority targets.
Technology Infrastructure
Modern sales requires sophisticated CRM systems, sample management platforms, and analytics capabilities. According to research on IT technology benefits in clinical trials, outsourcing strategy paired with robust technology improves compliance and quality outcomes.
Evaluate whether partners use industry-standard platforms (Salesforce, Veeva), provide real-time reporting dashboards, and integrate with your existing systems. Data visibility shouldn't decrease just because sales are outsourced.
Compliance Track Record
Request documentation of compliance training programs, audit results, and any regulatory citations. Partners with FDA warning letters or industry violations introduce unacceptable risk.
Ensure the partner maintains current PhRMA Code training, adverse event reporting procedures, and promotional review processes that align with your standards.
Financial Stability and Scale
Can the partner support your growth trajectory? A CSO struggling financially may lack resources for adequate training or technology investments. Conversely, very large organizations may treat smaller clients as low priority.
Review financial statements, client retention rates, and growth projections. Partners experiencing high turnover or client losses signal operational problems.

Critical Compliance Considerations
Regulatory compliance isn't negotiable in health sciences. Outsourcing creates additional complexity that requires proactive management.
FDA Oversight and Responsibility
Companies retain full regulatory responsibility even when sales activities are outsourced. FDA doesn't recognize outsourcing as a defense against promotional violations or adverse event reporting failures.
For facilities registered as outsourcing facilities under section 503B, FDA conducts inspections and issues warning letters when violations occur. Recent enforcement actions demonstrate the agency's commitment to holding both manufacturers and their partners accountable.
Promotional Material Review
All sales materials, presentations, and promotional content require thorough review before use. Establish joint review committees that include regulatory affairs, medical affairs, legal, and partner representatives.
Many companies require partners to submit all materials through formal MLR (Medical Legal Regulatory) review processes, with documented approvals before distribution.
Adverse Event Reporting
Sales reps often receive the first reports of product problems or adverse events. Outsourced teams must understand reporting obligations, timelines, and escalation procedures.
Build these requirements into training programs and audit them regularly. A single missed adverse event report can trigger FDA enforcement actions and legal liability.
Off-Label Promotion Risks
Healthcare professionals frequently ask about off-label uses. Reps must navigate these conversations carefully, providing only FDA-approved information while directing clinical questions to medical affairs.
Partners need clear protocols for handling off-label inquiries, documented training on permissible responses, and regular compliance monitoring.
Emerging Trends Reshaping Health Science Outsourcing
The outsourcing landscape continues evolving rapidly. Several trends are reshaping how companies approach these partnerships.
Chronic Disease Burden Driving Demand
According to WHO and UN data, chronic diseases (non-communicable diseases) are projected to account for 70% of all deaths globally by 2030. This creates sustained demand for new medical products with accelerated launch timelines—precisely the scenario where outsourced sales provides maximum advantage.
Digital and Hybrid Engagement Models
Telehealth adoption fundamentally changed healthcare sales. According to AMA data, while nearly 65-90% of physicians had the capability to provide telehealth in 2021, the actual consistent usage by physicians for consultations stabilized at around 25% of all patient visits by 2023-2024, though over 80% maintain the offering. Outsourcing partners increasingly blend traditional field sales with digital engagement capabilities.
Some 40% of patients indicate they'll continue using virtual care alternatives. This shift requires sales teams comfortable with virtual demonstrations, remote training, and digital relationship-building.
Value-Based Contracting Expertise
Healthcare systems increasingly demand outcomes-based pricing and risk-sharing arrangements. Sales teams need sophisticated understanding of health economics, real-world evidence, and value proposition development.
Outsourcing partners with dedicated value and access specialists help companies navigate these complex negotiations without building internal health economics teams.
Specialized Therapeutic Niches
As therapeutic areas become more specialized—gene therapies, precision oncology, rare diseases—sales expertise becomes harder to build internally. Outsourcing partners who invest in ultra-specialized training and certification provide access to capabilities that would be prohibitively expensive to develop in-house.
Implementation Best Practices
Successful outsourcing requires more than selecting the right partner. Implementation discipline determines whether the relationship delivers value.
Start with Pilot Programs
Rather than committing to large-scale outsourcing immediately, test the relationship through limited geography or specific product pilots. This allows both parties to refine processes, identify gaps, and build trust before scaling.
Define clear success metrics for the pilot—sales targets, relationship quality indicators, compliance audit results—and use these to guide expansion decisions.
Invest in Comprehensive Onboarding
Treat outsourced reps like internal employees during onboarding. Provide the same clinical training, product knowledge development, and company culture immersion.
Companies that skimp on training see it reflected in rep credibility and sales results. The investment pays dividends through better customer interactions and faster ramp times.
Establish Clear Communication Cadence
Weekly or biweekly sync meetings between internal teams and outsourced partners prevent misalignment. These should cover pipeline development, competitive intelligence, customer feedback, and tactical adjustments.
Don't rely solely on formal reporting. Regular informal communication builds relationships and surfaces issues before they become problems.
Create Joint Success Metrics
Align incentives by measuring what matters to both parties. Beyond sales volume, consider metrics like customer satisfaction scores, compliance audit results, relationship depth indicators, and strategic account penetration.
When partners share accountability for outcomes beyond just revenue, collaboration improves and short-term thinking decreases.
Conduct Regular Performance Reviews
Quarterly business reviews should assess performance against SLAs, identify improvement opportunities, and recalibrate priorities. These aren't punitive exercises—they're collaborative sessions to optimize the partnership.
Include recognition of successes alongside constructive feedback. Partners who feel valued as strategic collaborators deliver better results than those treated as interchangeable vendors.
Conclusion: Strategic Outsourcing Requires Strategic Planning
Health science sales outsourcing has matured from a tactical cost-reduction tool into a strategic capability that accelerates market entry, provides specialized expertise, and creates financial flexibility. The medical device outsourcing market's projected growth to USD 140.08 billion by 2032 reflects this fundamental shift.
But success isn't automatic. Companies that approach outsourcing strategically—selecting partners based on therapeutic expertise and compliance track records, investing in comprehensive training, establishing clear metrics, and treating partners as true collaborators—achieve significantly better results than those simply seeking the lowest cost provider.
The regulatory environment adds complexity that can't be delegated. FDA oversight, promotional compliance, and adverse event reporting remain the manufacturer's responsibility regardless of outsourcing arrangements. Partners must demonstrate not just sales capability, but regulatory sophistication and commitment to compliance.
For companies facing accelerated launch timelines, resource constraints, or expansion into new therapeutic areas, outsourcing provides a proven path forward. The key is approaching these partnerships with the same strategic rigor applied to internal capability development.
Ready to explore health science sales outsourcing for your organization? Start by conducting a thorough needs assessment identifying which sales functions deliver maximum value when outsourced, developing detailed partner selection criteria weighted toward therapeutic expertise and compliance, and designing pilot programs that test capabilities before full-scale commitments. The right partner can become a competitive advantage—choose deliberately.
Frequently Asked Questions
Topics
Related Blogs
Related Podcasts














