.avif)
.png)
Medical AI outsourcing enables healthcare organizations to access specialized AI expertise for developing diagnostic tools, predictive models, and patient care systems without building costly in-house teams. This guide covers regulatory compliance (FDA guidelines), vendor selection criteria, data security protocols, and implementation strategies to help hospitals, clinics, and medtech companies successfully outsource AI development while maintaining quality and patient safety.
Healthcare organizations are facing a talent crunch. The demand for AI engineers who understand both machine learning and medical regulations far exceeds the available workforce. Building an in-house team capable of developing FDA-compliant AI diagnostic tools can take years and cost millions.
That's where medical AI outsourcing comes in. Hospitals, diagnostic networks, pharmaceutical companies, and medtech startups are increasingly partnering with specialized development firms to accelerate innovation without drowning in overhead costs.
But here's the thing—outsourcing medical AI isn't like outsourcing a standard web application. The stakes are higher, the regulations tighter, and the complexity greater.
What Medical AI Outsourcing Actually Means
Medical AI outsourcing involves contracting external development teams to build, train, validate, or maintain artificial intelligence systems used in healthcare settings. These systems range from diagnostic imaging algorithms to predictive patient risk models.
The FDA encourages the development of innovative, safe, and effective medical devices, including devices that incorporate artificial intelligence. According to the FDA, AI and machine learning technologies have the potential to transform health care by deriving new and important insights from the vast amount of data generated during the delivery of health care every day.
Outsourced medical AI projects typically fall into several categories:
- Diagnostic AI systems: Image analysis for radiology, pathology, dermatology
- Predictive analytics: Patient risk stratification, readmission prediction, disease progression modeling
- Clinical decision support: Treatment recommendation engines, drug interaction alerts
- Administrative automation: Prior authorization processing, medical coding, scheduling optimization
- Remote monitoring: Wearable data analysis, continuous patient monitoring systems
The global healthcare IT market is projected to reach USD 1,834.3 billion by 2030 with an expected CAGR of 15.8%. This explosive growth creates both opportunities and pressures for healthcare organizations to adopt AI capabilities quickly.
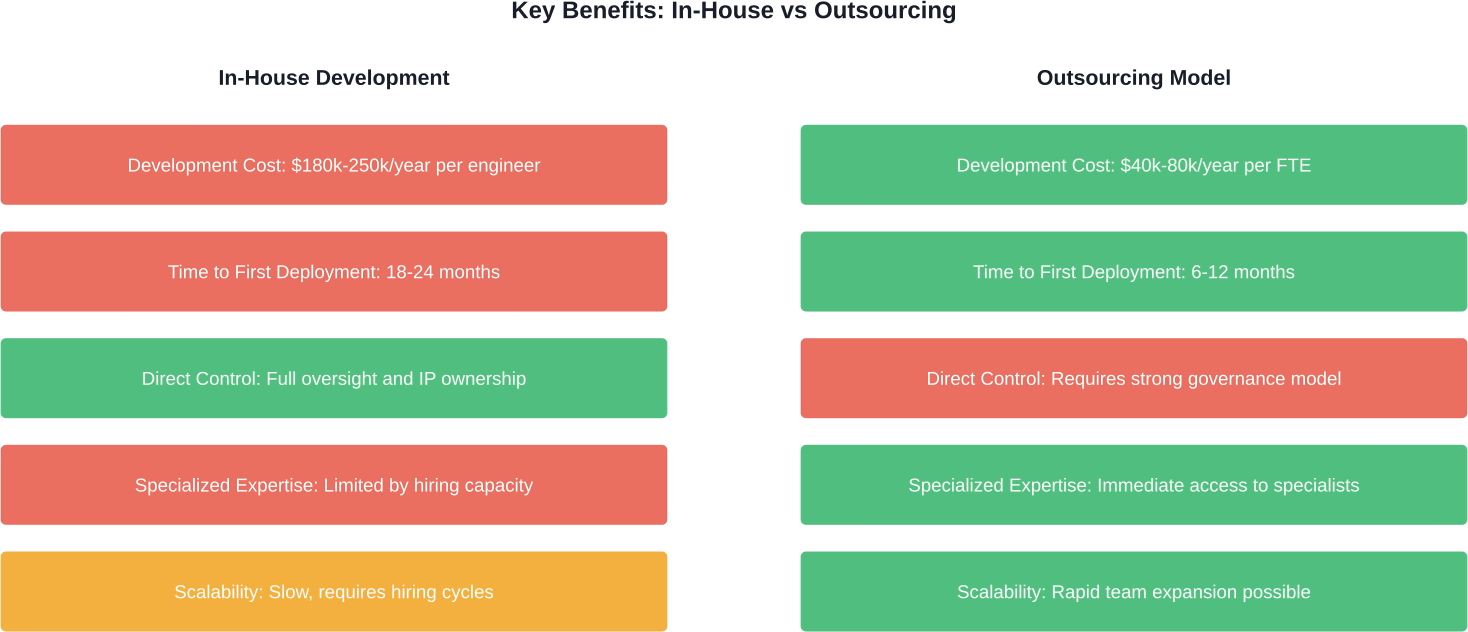
Why Healthcare Organizations Turn to Outsourcing
Cost efficiency tops the list of reasons hospitals and clinics outsource AI development. Senior AI engineers in North America typically command hourly rates of USD 120-200+ per hour. Outsourcing to partners in Eastern Europe or Asia can provide cost reductions compared to North American rates.
But cost isn't everything. Speed matters in healthcare technology.
Building an in-house AI team requires recruiting scarce talent, establishing infrastructure, creating training pipelines, and developing institutional knowledge around regulatory compliance. Building in-house AI teams typically requires significant time investment before delivering production-ready systems.
Outsourcing partners bring pre-existing expertise. They've already navigated FDA submission processes, implemented HIPAA-compliant data pipelines, and solved common technical challenges. According to industry reports, many enterprise-level companies now outsource at least one part of their AI projects.
Access to specialized skills represents another critical advantage. Medical AI requires interdisciplinary expertise spanning machine learning engineering, medical domain knowledge, regulatory compliance, clinical validation methodology, and healthcare data security protocols. Few organizations can maintain all these competencies internally.
Understanding FDA Requirements for AI Medical Devices
The FDA's regulatory framework shapes every medical AI outsourcing decision. In January 2025, the FDA issued draft guidance providing comprehensive recommendations for AI-enabled devices throughout the total product lifecycle. This represents the first guidance to address AI medical devices from development through post-market monitoring.
The FDA maintains an AI-Enabled Medical Device List that identifies AI-enabled medical devices authorized for marketing in the United States. Digital health innovators can reference this list to gain insights into the current device landscape and regulatory expectations.
When outsourcing medical AI development, organizations must ensure their external partners understand these regulatory pathways:
- 510(k) Premarket Notification: Required for devices substantially equivalent to existing legally marketed devices. Most AI diagnostic tools follow this pathway.
- De Novo Classification: For novel, low-to-moderate-risk devices without a predicate. Some innovative AI applications qualify for this streamlined process.
- Premarket Approval (PMA): Required for high-risk devices. Complex AI systems that directly control treatment typically need PMA.
The complex and dynamic processes involved in the development, deployment, use, and maintenance of AI technologies require specialized expertise. Outsourcing partners should demonstrate prior FDA submission experience and maintain documentation practices that support regulatory review.
Software as a Medical Device Considerations
The FDA categorizes many AI systems as Software as a Medical Device (SaMD). This classification carries specific requirements for validation, performance testing, and clinical evidence.
According to FDA guidance on artificial intelligence in Software as a Medical Device, manufacturers must address the iterative nature of AI algorithms. Traditional medical devices remain static post-approval, but AI systems can adapt and change based on new data.
Outsourcing agreements must specify how algorithm updates will be managed, validated, and documented to maintain regulatory compliance. This includes establishing protocols for continuous learning systems and defining when changes require new FDA submissions.
Data Security and Privacy Compliance
Medical AI development requires access to patient data. This creates significant security and privacy obligations that outsourcing partners must meet.
HIPAA compliance forms the baseline requirement for any vendor handling protected health information (PHI). Business Associate Agreements (BAAs) must be in place before any PHI transfer occurs. These agreements legally bind the outsourcing partner to HIPAA standards and establish liability frameworks.
But HIPAA represents just the starting point. Research from IEEE conferences on data security challenges in AI-enabled medical device software identifies several critical risk areas:
- Data transmission security during model training
- Cloud storage encryption and access controls
- De-identification techniques that preserve data utility while protecting privacy
- Audit trails documenting who accessed what data when
- Incident response procedures for data breaches
IEEE research on outsourcing artificial neural network learning of privacy-sensitive medical data to the cloud demonstrates that proper encryption and access controls can enable secure cloud-based AI development. However, implementation requires technical sophistication.
Organizations should require outsourcing partners to maintain SOC 2 Type II certification, demonstrating audited security controls. ISO 27001 certification provides additional assurance of information security management practices.
International Data Transfer Considerations
When outsourcing to international partners, data residency requirements complicate compliance. European GDPR regulations restrict PHI transfer outside the EU without adequate safeguards. Some U.S. healthcare systems have internal policies prohibiting offshore data storage.
Three approaches address these constraints:
- Synthetic data generation: Partners train initial models using synthetic datasets that mimic real patient data characteristics without containing actual PHI. Final validation occurs on real data within compliant environments.
- Federated learning: Models train across distributed datasets without centralizing patient data. The outsourcing partner develops the architecture while data remains in approved locations.
- On-premise deployment: Development teams work within the healthcare organization's secure environment, either on-site or through compliant remote access solutions.
Vendor Selection Framework
Choosing the right medical AI outsourcing partner determines project success. The selection process should evaluate technical capabilities, regulatory expertise, security practices, and cultural fit.
Request case studies demonstrating similar project complexity. Generic testimonials provide limited value—ask for specific technical challenges the vendor overcame and validation results achieved.
Reference checks matter more in medical AI than other outsourcing domains. Speak directly with previous healthcare clients about regulatory navigation, data security practices, and problem-solving approaches.
Evaluating Technical Capabilities
Technical assessments should go beyond résumé review. Request architecture diagrams from previous medical AI projects. Evaluate their approach to model validation, performance monitoring, and bias detection.
According to guidelines for contributors to medical artificial intelligence research published in medical informatics journals, comprehensive reporting standards are essential. Vendors should demonstrate familiarity with TRIPOD guidelines for predictive models, STARD standards for diagnostic accuracy, and CONSORT-AI extensions for clinical trials involving AI.
Ask candidates to explain their model evaluation metrics. Performance metrics should include the primary metric for model selection (such as area under the receiver operating characteristic curve for diagnostic systems) presented with statistical uncertainty measures like confidence intervals.

Build Your Medical AI Team Without the Hiring Bottleneck
Medical AI projects need more than strong models. They need engineers and support teams who understand healthcare workflows and stay long enough to see the roadmap through. NeoWork helps companies build dedicated remote teams for AI engineering, data operations, QA, and back-office roles that support regulated medical environments. With a 91% annualized retention rate, they focus on long-term teammates, not short-term churn. Their 3.2% candidate selectivity rate also means you are not sorting through unqualified applicants.
If you are planning a serious medical AI rollout and cannot afford hiring delays or constant turnover, talk to NeoWork and build a team that will still be there when your product reaches scale.
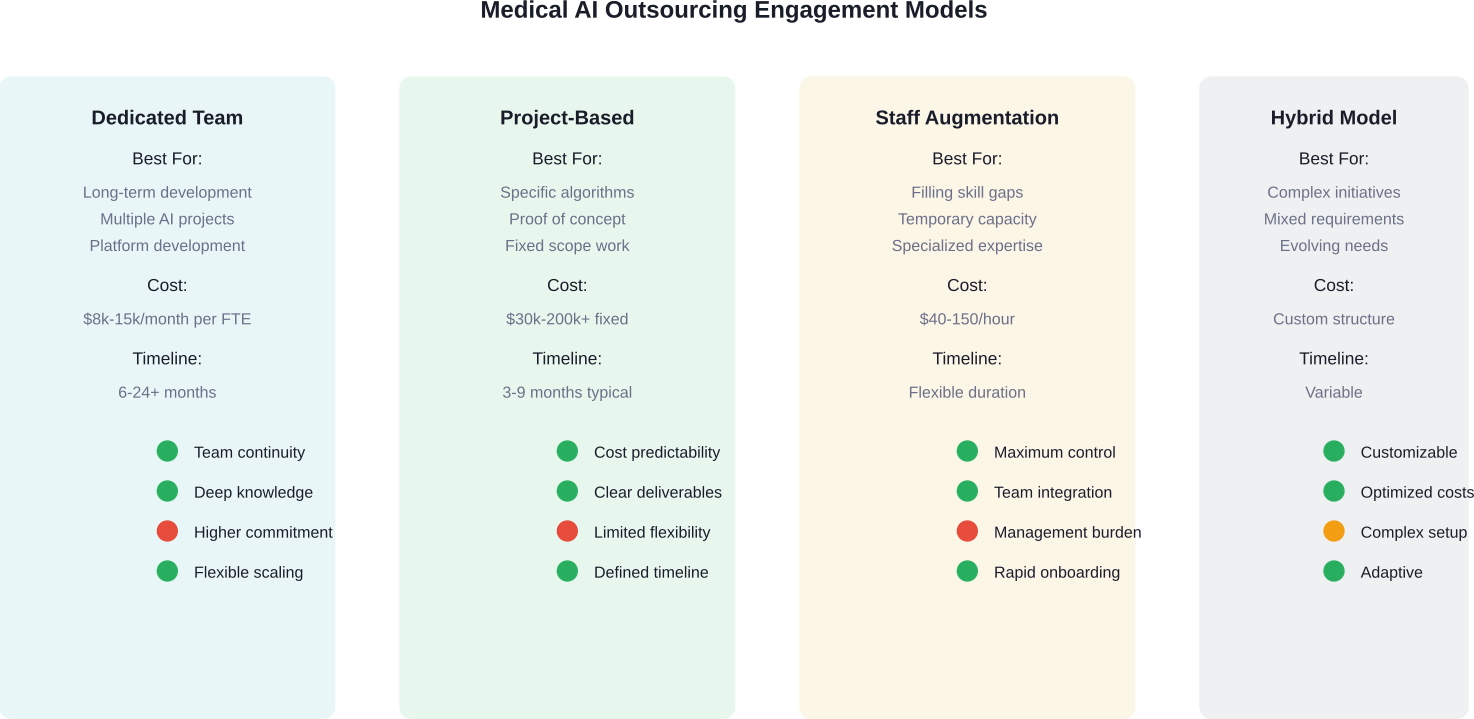
Common Outsourcing Models for Medical AI
Different engagement models suit different organizational needs and project types. Understanding these options helps structure effective partnerships.
- Dedicated team model: The vendor assembles a team exclusively focused on the client's projects. This model provides consistency and deep institutional knowledge but requires longer-term commitment. Monthly costs for dedicated teams vary based on location and expertise level.
- Project-based model: Defined scope, timeline, and deliverables with fixed or milestone-based pricing. Works well for discrete projects like developing a specific diagnostic algorithm. Provides cost predictability but less flexibility for scope changes.
- Staff augmentation: Individual specialists integrate into existing internal teams. Offers maximum control and cultural integration but requires strong internal project management. Outsourcing to international partners can provide cost reductions through location-based pricing models.
- Hybrid model: Combines elements of multiple approaches. For example, a dedicated core team handles ongoing development while subject matter experts provide consultation on specific challenges.
Implementation Best Practices
Successful medical AI outsourcing requires more than selecting the right vendor. Implementation practices determine whether partnerships deliver expected results.
Scoping and Requirements Definition
Medical AI projects fail most often due to ambiguous requirements rather than technical limitations. Scoping should define needs, compliance requirements, and data availability upfront to prevent timeline drift.
According to the FUTURE-AI international consensus guideline for trustworthy and deployable artificial intelligence in healthcare, recommendations that garnered high-level agreement (greater than 90%) emphasize transparent documentation of intended use, target populations, and clinical workflows.
Requirements documents should specify:
- Clinical context and intended use case
- Target performance metrics with acceptable ranges
- Data sources and availability timelines
- Regulatory pathway and submission requirements
- Integration points with existing systems
- Validation methodology and success criteria
Define measurable KPIs from day one. For diagnostic systems, this might include accuracy greater than 90% or latency less than one second. For predictive models, specify required area under the curve thresholds and acceptable false positive rates.
Data Preparation and Quality
Data preparation represents the biggest time impact in medical AI development. Cleaning, structuring, and verifying datasets typically consumes 40-60% of project timelines.
Outsourcing partners need well-documented data dictionaries explaining variable definitions, coding systems, and data collection contexts. Ambiguous or inconsistent data formats create cascading delays.
Establish data quality metrics before development begins. Missing data percentages, label accuracy rates, and class distribution balance all affect model performance. Addressing these issues early prevents late-stage rework.
Communication and Project Governance
Geographic and organizational distance creates communication challenges. Successful outsourcing relationships establish clear protocols for regular interaction.
Daily standups keep distributed teams aligned on priorities and blockers. Weekly sprint reviews demonstrate progress and enable course corrections. Monthly business reviews assess overall trajectory against milestones.
Documentation standards matter more with external teams. Architecture decisions, model selection rationale, validation results, and deployment procedures all require clear written records. This documentation becomes essential during FDA submissions and internal audits.
Assign dedicated internal stakeholders as single points of contact. Split accountability between multiple internal reviewers slows decision-making and creates confusion.
Critical Challenges and Risk Mitigation
Medical AI outsourcing introduces specific risks that standard software outsourcing doesn't face. Understanding and mitigating these challenges separates successful partnerships from failed ones.
Regulatory Compliance Gaps
Not all AI development firms understand healthcare regulatory requirements. Vendors without FDA experience may build technically impressive systems that can't pass regulatory review.
Mitigation strategies include requiring regulatory specialists on the vendor team, establishing checkpoints for FDA guidance alignment, and conducting mock pre-submission meetings to identify issues early.
The FDA's Medical Device Software Guidance Navigator provides a resource to help innovators identify relevant FDA guidance documents for their medical device software development and marketing submissions. Outsourcing partners should demonstrate familiarity with this resource and relevant guidance documents.
Clinical Validation Challenges
Technical validation on holdout datasets doesn't guarantee clinical utility. AI systems must demonstrate value in real clinical workflows with actual providers and patients.
Plan clinical validation early in project scoping. Identify validation sites, secure necessary institutional review board approvals, and establish clinical champion relationships before development completion.
Retrospective validation on historical data provides initial evidence but prospective studies carry more weight with regulators and clinicians. Budget additional time and resources for prospective validation.
Intellectual Property Considerations
IP ownership requires explicit contractual definition. Standard software development agreements may not address the unique aspects of AI systems including training data rights, model weights ownership, and derivative work provisions.
Contracts should specify that all work products including code, trained models, documentation, and technical knowledge belong to the healthcare organization. Work-for-hire clauses establish this ownership clearly.
Some vendors maintain platform components across multiple clients. Distinguish between proprietary platform elements the vendor retains and project-specific deliverables that transfer to the client.
Model Performance Degradation
AI models can degrade over time as clinical practices evolve, patient populations shift, or data collection methods change. This phenomenon, called data drift, requires ongoing monitoring and maintenance.
Establish performance monitoring systems before deployment. Track key metrics continuously and define thresholds that trigger model retraining or review.
Outsourcing agreements should include post-deployment support covering model monitoring, retraining, and updates. One-time development contracts leave organizations unable to maintain model performance.
Real-World Use Cases
Understanding how organizations successfully implement medical AI outsourcing provides practical insights beyond theoretical frameworks:
- Radiology diagnostic support: A regional hospital network outsourced development of a chest X-ray pneumonia detection system. The vendor provided ML engineers and medical imaging specialists who worked with the hospital's radiologists to label training data, validate model outputs, and integrate the system into existing PACS workflows. The project achieved 94% sensitivity for pneumonia detection within eight months, submitted 510(k) documentation, and gained FDA clearance.
- Patient risk stratification: A health insurance company partnered with an outsourcing firm to develop predictive models identifying high-risk members for care management programs. The vendor built models using claims data, electronic health record extracts, and social determinants data. The system identifies patients at elevated risk for hospital readmission with 82% accuracy, enabling proactive interventions that reduced readmissions by 17%.
- Pathology image analysis: A pathology laboratory outsourced development of an AI system for detecting cancer in biopsy samples. The vendor specialized in computer vision and digital pathology. Development included creating annotation tools for pathologists, training deep learning models on over 50,000 annotated slides, and validating against expert consensus diagnoses. The system now assists pathologists by flagging suspicious regions for detailed review.
- Clinical decision support: A specialty pharmaceutical company engaged an outsourcing partner to develop treatment recommendation algorithms for rare diseases. The vendor's medical informaticists and data scientists built natural language processing systems to extract relevant information from clinical notes, combined this with structured data, and generated evidence-based treatment suggestions. The system supports clinicians in selecting appropriate therapies based on patient characteristics and latest research.
Cost Structure and Budget Planning
Medical AI outsourcing costs vary significantly based on project complexity, vendor location, engagement model, and regulatory requirements.
These ranges assume mid-complexity projects with established data sources. Novel algorithms, challenging regulatory pathways, or extensive clinical validation increase costs significantly.
Hidden costs often surprise organizations new to medical AI outsourcing. Internal stakeholder time for requirements definition, data access approvals, clinical validation, and regulatory review represents substantial opportunity cost. Budget for dedicated internal resources, not just vendor fees.
FDA submission fees add additional costs. The standard fee for a 510(k) submission is $24,335 (or $6,084 for small businesses).
Future Trends Shaping Medical AI Outsourcing
The medical AI outsourcing landscape continues evolving. Several trends will shape how healthcare organizations approach external partnerships in coming years.
- Foundation model specialization: Large language models and vision transformers are being fine-tuned for medical applications. Outsourcing partners increasingly offer pre-trained foundation models that require less training data and development time for specific use cases. This shifts development from training models from scratch to adapting and validating existing architectures.
- Continuous validation systems: Traditional one-time validation approaches don't address the dynamic nature of AI systems. Outsourcing providers are developing continuous monitoring and validation frameworks that assess model performance in real-time, detect degradation, and trigger retraining workflows automatically.
- Federated learning adoption: Privacy-preserving techniques enabling model training across distributed datasets without centralizing patient information are moving from research to production. This allows multi-site collaborations and international partnerships while maintaining data sovereignty.
- Regulatory harmonization: International standards bodies are working toward harmonized medical AI regulations. The FUTURE-AI framework and similar efforts aim to create consistent development and validation standards across jurisdictions, simplifying global deployment.
- Edge deployment: Moving AI inference from cloud servers to edge devices reduces latency, improves privacy, and enables offline operation. Outsourcing partners with expertise in model compression, edge optimization, and embedded systems are increasingly valuable.
Taking the Next Step in Medical AI Outsourcing
Medical AI outsourcing offers healthcare organizations a pragmatic path to advanced capabilities without the time and expense of building specialized internal teams. The global healthcare IT market's rapid expansion to USD 1,834.3 billion by 2030 reflects how deeply AI is transforming medical practice.
Success requires more than finding the cheapest developers. It demands partners who understand both machine learning and the unique challenges of healthcare—regulatory compliance, clinical validation, patient privacy, and the life-and-death stakes of medical applications.
The organizations achieving the best outcomes approach outsourcing strategically. They invest time in vendor evaluation, define requirements clearly, maintain active project involvement, and plan for long-term system maintenance rather than treating AI as a one-time development effort.
Real talk: Medical AI outsourcing isn't the right choice for every organization or every project. Small-scale internal pilots, highly sensitive proprietary algorithms, or projects requiring minute-by-minute collaboration may warrant in-house development. But for most healthcare organizations, partnering with specialized external teams accelerates innovation while managing costs.
Start by defining the specific clinical problem AI should solve. Work backward from that to data availability, regulatory pathway, and implementation requirements. Use these details to evaluate potential partners against the criteria outlined above.
The healthcare organizations leading AI adoption aren't necessarily the largest or best-funded. They're the ones who recognized they couldn't build every capability internally and found the right partners to fill gaps.
Ready to explore medical AI outsourcing for your organization? Begin with a focused pilot project addressing a specific clinical challenge. Use that experience to refine processes, evaluate vendor performance, and build institutional knowledge before expanding to larger initiatives.
Frequently Asked Questions
Topics
Medical AI Outsourcing Guide 2026: Complete Roadmap
Medical AI outsourcing enables healthcare organizations to access specialized AI expertise for developing diagnostic tools, predictive models, and patient care systems without building costly in-house teams. This guide covers regulatory compliance (FDA guidelines), vendor selection criteria, data security protocols, and implementation strategies to help hospitals, clinics, and medtech companies successfully outsource AI development while maintaining quality and patient safety.
Healthcare organizations are facing a talent crunch. The demand for AI engineers who understand both machine learning and medical regulations far exceeds the available workforce. Building an in-house team capable of developing FDA-compliant AI diagnostic tools can take years and cost millions.
That's where medical AI outsourcing comes in. Hospitals, diagnostic networks, pharmaceutical companies, and medtech startups are increasingly partnering with specialized development firms to accelerate innovation without drowning in overhead costs.
But here's the thing—outsourcing medical AI isn't like outsourcing a standard web application. The stakes are higher, the regulations tighter, and the complexity greater.
What Medical AI Outsourcing Actually Means
Medical AI outsourcing involves contracting external development teams to build, train, validate, or maintain artificial intelligence systems used in healthcare settings. These systems range from diagnostic imaging algorithms to predictive patient risk models.
The FDA encourages the development of innovative, safe, and effective medical devices, including devices that incorporate artificial intelligence. According to the FDA, AI and machine learning technologies have the potential to transform health care by deriving new and important insights from the vast amount of data generated during the delivery of health care every day.
Outsourced medical AI projects typically fall into several categories:
- Diagnostic AI systems: Image analysis for radiology, pathology, dermatology
- Predictive analytics: Patient risk stratification, readmission prediction, disease progression modeling
- Clinical decision support: Treatment recommendation engines, drug interaction alerts
- Administrative automation: Prior authorization processing, medical coding, scheduling optimization
- Remote monitoring: Wearable data analysis, continuous patient monitoring systems
The global healthcare IT market is projected to reach USD 1,834.3 billion by 2030 with an expected CAGR of 15.8%. This explosive growth creates both opportunities and pressures for healthcare organizations to adopt AI capabilities quickly.
Why Healthcare Organizations Turn to Outsourcing
Cost efficiency tops the list of reasons hospitals and clinics outsource AI development. Senior AI engineers in North America typically command hourly rates of USD 120-200+ per hour. Outsourcing to partners in Eastern Europe or Asia can provide cost reductions compared to North American rates.
But cost isn't everything. Speed matters in healthcare technology.
Building an in-house AI team requires recruiting scarce talent, establishing infrastructure, creating training pipelines, and developing institutional knowledge around regulatory compliance. Building in-house AI teams typically requires significant time investment before delivering production-ready systems.
Outsourcing partners bring pre-existing expertise. They've already navigated FDA submission processes, implemented HIPAA-compliant data pipelines, and solved common technical challenges. According to industry reports, many enterprise-level companies now outsource at least one part of their AI projects.
Access to specialized skills represents another critical advantage. Medical AI requires interdisciplinary expertise spanning machine learning engineering, medical domain knowledge, regulatory compliance, clinical validation methodology, and healthcare data security protocols. Few organizations can maintain all these competencies internally.

Understanding FDA Requirements for AI Medical Devices
The FDA's regulatory framework shapes every medical AI outsourcing decision. In January 2025, the FDA issued draft guidance providing comprehensive recommendations for AI-enabled devices throughout the total product lifecycle. This represents the first guidance to address AI medical devices from development through post-market monitoring.
The FDA maintains an AI-Enabled Medical Device List that identifies AI-enabled medical devices authorized for marketing in the United States. Digital health innovators can reference this list to gain insights into the current device landscape and regulatory expectations.
When outsourcing medical AI development, organizations must ensure their external partners understand these regulatory pathways:
- 510(k) Premarket Notification: Required for devices substantially equivalent to existing legally marketed devices. Most AI diagnostic tools follow this pathway.
- De Novo Classification: For novel, low-to-moderate-risk devices without a predicate. Some innovative AI applications qualify for this streamlined process.
- Premarket Approval (PMA): Required for high-risk devices. Complex AI systems that directly control treatment typically need PMA.
The complex and dynamic processes involved in the development, deployment, use, and maintenance of AI technologies require specialized expertise. Outsourcing partners should demonstrate prior FDA submission experience and maintain documentation practices that support regulatory review.
Software as a Medical Device Considerations
The FDA categorizes many AI systems as Software as a Medical Device (SaMD). This classification carries specific requirements for validation, performance testing, and clinical evidence.
According to FDA guidance on artificial intelligence in Software as a Medical Device, manufacturers must address the iterative nature of AI algorithms. Traditional medical devices remain static post-approval, but AI systems can adapt and change based on new data.
Outsourcing agreements must specify how algorithm updates will be managed, validated, and documented to maintain regulatory compliance. This includes establishing protocols for continuous learning systems and defining when changes require new FDA submissions.
Data Security and Privacy Compliance
Medical AI development requires access to patient data. This creates significant security and privacy obligations that outsourcing partners must meet.
HIPAA compliance forms the baseline requirement for any vendor handling protected health information (PHI). Business Associate Agreements (BAAs) must be in place before any PHI transfer occurs. These agreements legally bind the outsourcing partner to HIPAA standards and establish liability frameworks.
But HIPAA represents just the starting point. Research from IEEE conferences on data security challenges in AI-enabled medical device software identifies several critical risk areas:
- Data transmission security during model training
- Cloud storage encryption and access controls
- De-identification techniques that preserve data utility while protecting privacy
- Audit trails documenting who accessed what data when
- Incident response procedures for data breaches
IEEE research on outsourcing artificial neural network learning of privacy-sensitive medical data to the cloud demonstrates that proper encryption and access controls can enable secure cloud-based AI development. However, implementation requires technical sophistication.
Organizations should require outsourcing partners to maintain SOC 2 Type II certification, demonstrating audited security controls. ISO 27001 certification provides additional assurance of information security management practices.
International Data Transfer Considerations
When outsourcing to international partners, data residency requirements complicate compliance. European GDPR regulations restrict PHI transfer outside the EU without adequate safeguards. Some U.S. healthcare systems have internal policies prohibiting offshore data storage.
Three approaches address these constraints:
- Synthetic data generation: Partners train initial models using synthetic datasets that mimic real patient data characteristics without containing actual PHI. Final validation occurs on real data within compliant environments.
- Federated learning: Models train across distributed datasets without centralizing patient data. The outsourcing partner develops the architecture while data remains in approved locations.
- On-premise deployment: Development teams work within the healthcare organization's secure environment, either on-site or through compliant remote access solutions.
Vendor Selection Framework
Choosing the right medical AI outsourcing partner determines project success. The selection process should evaluate technical capabilities, regulatory expertise, security practices, and cultural fit.
Request case studies demonstrating similar project complexity. Generic testimonials provide limited value—ask for specific technical challenges the vendor overcame and validation results achieved.
Reference checks matter more in medical AI than other outsourcing domains. Speak directly with previous healthcare clients about regulatory navigation, data security practices, and problem-solving approaches.
Evaluating Technical Capabilities
Technical assessments should go beyond résumé review. Request architecture diagrams from previous medical AI projects. Evaluate their approach to model validation, performance monitoring, and bias detection.
According to guidelines for contributors to medical artificial intelligence research published in medical informatics journals, comprehensive reporting standards are essential. Vendors should demonstrate familiarity with TRIPOD guidelines for predictive models, STARD standards for diagnostic accuracy, and CONSORT-AI extensions for clinical trials involving AI.
Ask candidates to explain their model evaluation metrics. Performance metrics should include the primary metric for model selection (such as area under the receiver operating characteristic curve for diagnostic systems) presented with statistical uncertainty measures like confidence intervals.

Build Your Medical AI Team Without the Hiring Bottleneck
Medical AI projects need more than strong models. They need engineers and support teams who understand healthcare workflows and stay long enough to see the roadmap through. NeoWork helps companies build dedicated remote teams for AI engineering, data operations, QA, and back-office roles that support regulated medical environments. With a 91% annualized retention rate, they focus on long-term teammates, not short-term churn. Their 3.2% candidate selectivity rate also means you are not sorting through unqualified applicants.
If you are planning a serious medical AI rollout and cannot afford hiring delays or constant turnover, talk to NeoWork and build a team that will still be there when your product reaches scale.
Common Outsourcing Models for Medical AI
Different engagement models suit different organizational needs and project types. Understanding these options helps structure effective partnerships.
- Dedicated team model: The vendor assembles a team exclusively focused on the client's projects. This model provides consistency and deep institutional knowledge but requires longer-term commitment. Monthly costs for dedicated teams vary based on location and expertise level.
- Project-based model: Defined scope, timeline, and deliverables with fixed or milestone-based pricing. Works well for discrete projects like developing a specific diagnostic algorithm. Provides cost predictability but less flexibility for scope changes.
- Staff augmentation: Individual specialists integrate into existing internal teams. Offers maximum control and cultural integration but requires strong internal project management. Outsourcing to international partners can provide cost reductions through location-based pricing models.
- Hybrid model: Combines elements of multiple approaches. For example, a dedicated core team handles ongoing development while subject matter experts provide consultation on specific challenges.

Implementation Best Practices
Successful medical AI outsourcing requires more than selecting the right vendor. Implementation practices determine whether partnerships deliver expected results.
Scoping and Requirements Definition
Medical AI projects fail most often due to ambiguous requirements rather than technical limitations. Scoping should define needs, compliance requirements, and data availability upfront to prevent timeline drift.
According to the FUTURE-AI international consensus guideline for trustworthy and deployable artificial intelligence in healthcare, recommendations that garnered high-level agreement (greater than 90%) emphasize transparent documentation of intended use, target populations, and clinical workflows.
Requirements documents should specify:
- Clinical context and intended use case
- Target performance metrics with acceptable ranges
- Data sources and availability timelines
- Regulatory pathway and submission requirements
- Integration points with existing systems
- Validation methodology and success criteria
Define measurable KPIs from day one. For diagnostic systems, this might include accuracy greater than 90% or latency less than one second. For predictive models, specify required area under the curve thresholds and acceptable false positive rates.
Data Preparation and Quality
Data preparation represents the biggest time impact in medical AI development. Cleaning, structuring, and verifying datasets typically consumes 40-60% of project timelines.
Outsourcing partners need well-documented data dictionaries explaining variable definitions, coding systems, and data collection contexts. Ambiguous or inconsistent data formats create cascading delays.
Establish data quality metrics before development begins. Missing data percentages, label accuracy rates, and class distribution balance all affect model performance. Addressing these issues early prevents late-stage rework.
Communication and Project Governance
Geographic and organizational distance creates communication challenges. Successful outsourcing relationships establish clear protocols for regular interaction.
Daily standups keep distributed teams aligned on priorities and blockers. Weekly sprint reviews demonstrate progress and enable course corrections. Monthly business reviews assess overall trajectory against milestones.
Documentation standards matter more with external teams. Architecture decisions, model selection rationale, validation results, and deployment procedures all require clear written records. This documentation becomes essential during FDA submissions and internal audits.
Assign dedicated internal stakeholders as single points of contact. Split accountability between multiple internal reviewers slows decision-making and creates confusion.
Critical Challenges and Risk Mitigation
Medical AI outsourcing introduces specific risks that standard software outsourcing doesn't face. Understanding and mitigating these challenges separates successful partnerships from failed ones.
Regulatory Compliance Gaps
Not all AI development firms understand healthcare regulatory requirements. Vendors without FDA experience may build technically impressive systems that can't pass regulatory review.
Mitigation strategies include requiring regulatory specialists on the vendor team, establishing checkpoints for FDA guidance alignment, and conducting mock pre-submission meetings to identify issues early.
The FDA's Medical Device Software Guidance Navigator provides a resource to help innovators identify relevant FDA guidance documents for their medical device software development and marketing submissions. Outsourcing partners should demonstrate familiarity with this resource and relevant guidance documents.
Clinical Validation Challenges
Technical validation on holdout datasets doesn't guarantee clinical utility. AI systems must demonstrate value in real clinical workflows with actual providers and patients.
Plan clinical validation early in project scoping. Identify validation sites, secure necessary institutional review board approvals, and establish clinical champion relationships before development completion.
Retrospective validation on historical data provides initial evidence but prospective studies carry more weight with regulators and clinicians. Budget additional time and resources for prospective validation.
Intellectual Property Considerations
IP ownership requires explicit contractual definition. Standard software development agreements may not address the unique aspects of AI systems including training data rights, model weights ownership, and derivative work provisions.
Contracts should specify that all work products including code, trained models, documentation, and technical knowledge belong to the healthcare organization. Work-for-hire clauses establish this ownership clearly.
Some vendors maintain platform components across multiple clients. Distinguish between proprietary platform elements the vendor retains and project-specific deliverables that transfer to the client.
Model Performance Degradation
AI models can degrade over time as clinical practices evolve, patient populations shift, or data collection methods change. This phenomenon, called data drift, requires ongoing monitoring and maintenance.
Establish performance monitoring systems before deployment. Track key metrics continuously and define thresholds that trigger model retraining or review.
Outsourcing agreements should include post-deployment support covering model monitoring, retraining, and updates. One-time development contracts leave organizations unable to maintain model performance.
Real-World Use Cases
Understanding how organizations successfully implement medical AI outsourcing provides practical insights beyond theoretical frameworks:
- Radiology diagnostic support: A regional hospital network outsourced development of a chest X-ray pneumonia detection system. The vendor provided ML engineers and medical imaging specialists who worked with the hospital's radiologists to label training data, validate model outputs, and integrate the system into existing PACS workflows. The project achieved 94% sensitivity for pneumonia detection within eight months, submitted 510(k) documentation, and gained FDA clearance.
- Patient risk stratification: A health insurance company partnered with an outsourcing firm to develop predictive models identifying high-risk members for care management programs. The vendor built models using claims data, electronic health record extracts, and social determinants data. The system identifies patients at elevated risk for hospital readmission with 82% accuracy, enabling proactive interventions that reduced readmissions by 17%.
- Pathology image analysis: A pathology laboratory outsourced development of an AI system for detecting cancer in biopsy samples. The vendor specialized in computer vision and digital pathology. Development included creating annotation tools for pathologists, training deep learning models on over 50,000 annotated slides, and validating against expert consensus diagnoses. The system now assists pathologists by flagging suspicious regions for detailed review.
- Clinical decision support: A specialty pharmaceutical company engaged an outsourcing partner to develop treatment recommendation algorithms for rare diseases. The vendor's medical informaticists and data scientists built natural language processing systems to extract relevant information from clinical notes, combined this with structured data, and generated evidence-based treatment suggestions. The system supports clinicians in selecting appropriate therapies based on patient characteristics and latest research.
Cost Structure and Budget Planning
Medical AI outsourcing costs vary significantly based on project complexity, vendor location, engagement model, and regulatory requirements.
These ranges assume mid-complexity projects with established data sources. Novel algorithms, challenging regulatory pathways, or extensive clinical validation increase costs significantly.
Hidden costs often surprise organizations new to medical AI outsourcing. Internal stakeholder time for requirements definition, data access approvals, clinical validation, and regulatory review represents substantial opportunity cost. Budget for dedicated internal resources, not just vendor fees.
FDA submission fees add additional costs. The standard fee for a 510(k) submission is $24,335 (or $6,084 for small businesses).
Future Trends Shaping Medical AI Outsourcing
The medical AI outsourcing landscape continues evolving. Several trends will shape how healthcare organizations approach external partnerships in coming years.
- Foundation model specialization: Large language models and vision transformers are being fine-tuned for medical applications. Outsourcing partners increasingly offer pre-trained foundation models that require less training data and development time for specific use cases. This shifts development from training models from scratch to adapting and validating existing architectures.
- Continuous validation systems: Traditional one-time validation approaches don't address the dynamic nature of AI systems. Outsourcing providers are developing continuous monitoring and validation frameworks that assess model performance in real-time, detect degradation, and trigger retraining workflows automatically.
- Federated learning adoption: Privacy-preserving techniques enabling model training across distributed datasets without centralizing patient information are moving from research to production. This allows multi-site collaborations and international partnerships while maintaining data sovereignty.
- Regulatory harmonization: International standards bodies are working toward harmonized medical AI regulations. The FUTURE-AI framework and similar efforts aim to create consistent development and validation standards across jurisdictions, simplifying global deployment.
- Edge deployment: Moving AI inference from cloud servers to edge devices reduces latency, improves privacy, and enables offline operation. Outsourcing partners with expertise in model compression, edge optimization, and embedded systems are increasingly valuable.
Taking the Next Step in Medical AI Outsourcing
Medical AI outsourcing offers healthcare organizations a pragmatic path to advanced capabilities without the time and expense of building specialized internal teams. The global healthcare IT market's rapid expansion to USD 1,834.3 billion by 2030 reflects how deeply AI is transforming medical practice.
Success requires more than finding the cheapest developers. It demands partners who understand both machine learning and the unique challenges of healthcare—regulatory compliance, clinical validation, patient privacy, and the life-and-death stakes of medical applications.
The organizations achieving the best outcomes approach outsourcing strategically. They invest time in vendor evaluation, define requirements clearly, maintain active project involvement, and plan for long-term system maintenance rather than treating AI as a one-time development effort.
Real talk: Medical AI outsourcing isn't the right choice for every organization or every project. Small-scale internal pilots, highly sensitive proprietary algorithms, or projects requiring minute-by-minute collaboration may warrant in-house development. But for most healthcare organizations, partnering with specialized external teams accelerates innovation while managing costs.
Start by defining the specific clinical problem AI should solve. Work backward from that to data availability, regulatory pathway, and implementation requirements. Use these details to evaluate potential partners against the criteria outlined above.
The healthcare organizations leading AI adoption aren't necessarily the largest or best-funded. They're the ones who recognized they couldn't build every capability internally and found the right partners to fill gaps.
Ready to explore medical AI outsourcing for your organization? Begin with a focused pilot project addressing a specific clinical challenge. Use that experience to refine processes, evaluate vendor performance, and build institutional knowledge before expanding to larger initiatives.
Frequently Asked Questions
Topics
Related Blogs
Related Podcasts














